Carbon To Oxygen Bond Length
Bond Order and Lengths
- Page ID
- 1982
Bond order is the number of chemical bonds between a pair of atoms and indicates the stability of a bond. For instance, in diatomic nitrogen, N≡Due north, the bail order is 3; in acetylene, H−C≡C−H, the carbon-carbon bond gild is also 3, and the C−H bond gild is 1. Bond order and bond length indicate the type and forcefulness of covalent bonds between atoms. Bond order and length are inversely proportional to each other: when bond social club is increased, bond length is decreased.
Introduction
Chemistry deals with the way in which subatomic particles bond together to class atoms. Chemical science besides focuses on the fashion in which atoms bail together to form molecules. In the diminutive structure, electrons surround the atomic nucleus in regions called orbitals. Each orbital beat tin concur a certain number of electrons. When the nearest orbital shell is total, new electrons outset to assemble in the next orbital shell out from the nucleus, and continue until that beat out is too total. The collection of electrons continues in ever widening orbital shells as larger atoms have more electrons than smaller atoms. When 2 atoms bond to course a molecule, their electrons bail them together past mixing into openings in each others' orbital shells. As with the collection of electrons by the cantlet, the formation of bonds past the molecule starts at the nearest available orbital shell opening and expand outward.
Bond Order
Bail order is the number of bonding pairs of electrons betwixt two atoms. In a covalent bond between two atoms, a single bond has a bond society of 1, a double bond has a bail order of 2, a triple bond has a bond club of 3, and so on. To determine the bond order between two covalently bonded atoms, follow these steps:
- Depict the Lewis structure.
- Determine the blazon of bonds betwixt the 2 atoms.
- 0: No bond
- one: Single bond
- 2: double bond
- 3: triple bond
If the bond club is cypher, the molecule cannot course. The higher bond orders indicate greater stability for the new molecule. In molecules that take resonance bonding, the bond society does non need to be an integer.
Example \(\PageIndex{1}\): \(CN^-\)
Determine the bail order for cyanide, CN-.
Solution
i) Draw the Lewis structure.
ii) Determine the type of bail betwixt the two atoms.
Considering there are iii dashes, the bail is a triple bond. A triple bond corresponds to a bail order of 3.
Example \(\PageIndex{2}\): \(H_2\)
Determine the bond order for hydrogen gas, H2.
Solution
1) Draw the Lewis structure.
![]()
ii) Determine the type of bond between the two atoms.
There is simply one pair of shared electrons (or nuance), indicating is a unmarried bond, with a bond order of 1.
Polyatomic molecules
If at that place are more than two atoms in the molecule, follow these steps to determine the bail order:
- Depict the Lewis structure.
- Count the total number of bonds.
- Count the number of bail groups between individual atoms.
- Divide the number of bonds between atoms by the total number of bond groups in the molecule.
Example \(\PageIndex{three}\): \(NO_3^-\)
Determine the bail club for nitrate, \(NO_3^-\).
Solution
1) Draw the Lewis structure.
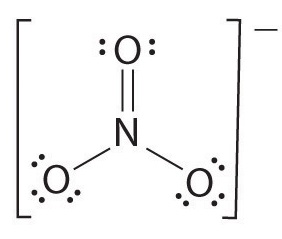
2) Count the full number of bonds.
four
The full number of bonds is 4.
3) Count the number of bond groups between individual atoms.
three
The number of bond groups between individual atoms is three.
4) Separate the number of bonds between individual atoms by the total number of bonds.
\[\dfrac{4}{three}= 1.33 \]
The bail gild is 1.33
Example \(\PageIndex{iv}\): \(NO^+_2\)
Determine the bond order for nitronium ion: \(NO_2^+\).
Solution
1) Draw the Lewis Construction.
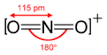
2) Count the full number of bonds.
4
The total number of bonds is four.
3) Count the number of bond groups betwixt individual atoms.
2
The number of bond groups betwixt atoms is ii.
4) Divide the bond groups between private atoms by the total number of bonds.
\[\frac{4}{ii} = 2\]
The bail lodge is 2.
A high bond guild indicates more attraction between electrons. A higher bail order also means that the atoms are held together more than tightly. With a lower bond order, in that location is less attraction between electrons and this causes the atoms to be held together more loosely. Bail order besides indicates the stability of the bond. The college the bond order, the more electrons property the atoms together, and therefore the greater the stability.
Trends in the Periodic Table
Bond order increases beyond a menses and decreases downward a group.
Bond Length
Bond length is defined as the altitude between the centers of two covalently bonded atoms. The length of the bond is determined by the number of bonded electrons (the bond club). The college the bond guild, the stronger the pull between the 2 atoms and the shorter the bond length. Generally, the length of the bail betwixt two atoms is approximately the sum of the covalent radii of the two atoms. Bond length is reported in picometers. Therefore, bond length increases in the post-obit society: triple bail < double bond < single bond.
To notice the bond length, follow these steps:
- Draw the Lewis structure.
- Look up the nautical chart below for the radii for the corresponding bond.
- Find the sum of the two radii.
iv
Determine the carbon-to-chlorine bond length in CCl4.
Solution
Using Table A3, a C single bond has a length of 75 picometers and that a Cl single bond has a length of 99 picometers. When added together, the bond length of a C-Cl bond is approximately 174 picometers.
ii
Decide the carbon-oxygen bond length in COtwo.
Solution
Using Table A3, we see that a C double bond has a length of 67 picometers and that an O double bail has a length of 57 picometers. When added together, the bond length of a C=O bond is approximately 124 picometers.
Trends in the Periodic Table
Because the bond length is proportional to the diminutive radius, the bail length trends in the periodic table follow the aforementioned trends as atomic radii: bond length decreases across a menses and increases downward a group.
Problems
- What is the bond order of \(O_2\)?
- What is the bail order of \(NO_3^-\)?
- What is the carbon-nitrogen bond length in \(HCN\)?
- Is the carbon-to-oxygen bond length greater in \(CO_2\) or \(CO\)?
- What is the nitrogen-fluoride bond length in \(NF_3\)?
Solutions
1. First, write the Lewis construction for \(O_2\).
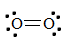
At that place is a double bond between the two oxygen atoms; therefore, the bond order of the molecule is two.
two. The Lewis structure for NO3 - is given below:
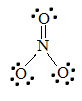
To detect the bond club of this molecule, take the average of the bond orders. Northward=O has a bond society of two, and both Due north-O bonds have a bond lodge of one. Adding these together and dividing by the number of bonds (3) reveals that the bail club of nitrate is ane.33.
3. To observe the carbon-nitrogen bond length in HCN, describe the Lewis structure of HCN.
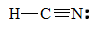
The bond between carbon and nitrogen is a triple bond, and a triple bail between carbon and nitrogen has a bail length of approximately 60 + 54 =114 pm.
iv. From the Lewis structures for CO2 and CO, at that place is a double bail between the carbon and oxygen in CO2 and a triple bond betwixt the carbon and oxygen in CO.

Referring to the table in a higher place, a double bond betwixt carbon and oxygen has a bond length of approximately 67 + 57 = 124 pm and a triple bail between carbon and oxygen has a bond length of approximately 60 + 53 =113 pm. Therefore, the bond length is greater in CO2.
Another method makes use of the fact that the more electron bonds between the atoms, the tighter the electrons are pulling the atoms together. Therefore, the bond length is greater in CO2.
five. To find the nitrogen-to-fluorine bond length in NF3, depict the Lewis structure.
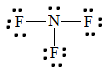
The bond betwixt fluorine and nitrogen is a single bond. From the table above, a unmarried bond between fluorine and nitrogen has a bond length of approximately 64 + 71 =135 pm.
References
- Campbell, Neil A., Brad Williamson, and Robin J. Heyden. Biological science: Exploring Life. Boston, Massachusetts: Pearson Prentice Hall, 2006.
- Petrucci, Ralph H., Harwood, William S., Herring, F. Chiliad., and Madura Jeffrey D. General Chemistry: Principles & Modern Applications. 9th Ed. New Bailiwick of jersey: Pearson Education, Inc., 2007. Print.
- Cordero, Beatriz, Verónica Gómez, Ana Eastward. Platero-Prats, Marc Revés, Jorge Echeverría, Eduard Cremades, Flavia Barragán and Santiago Alvarez. Dalton's Transactions." Covalent radii revisited 2008:
- Pekka Pyykkö and Michiko Atsumi, Chem. Eur. J. Molecular Double-Bond Covalent Radii for Elements Li–E112 2009
Contributors and Attributions
- Wikihow.com
- Bearding
Carbon To Oxygen Bond Length,
Source: https://chem.libretexts.org/Bookshelves/Physical_and_Theoretical_Chemistry_Textbook_Maps/Supplemental_Modules_%28Physical_and_Theoretical_Chemistry%29/Chemical_Bonding/Fundamentals_of_Chemical_Bonding/Bond_Order_and_Lengths
Posted by: williamsstectint98.blogspot.com


0 Response to "Carbon To Oxygen Bond Length"
Post a Comment